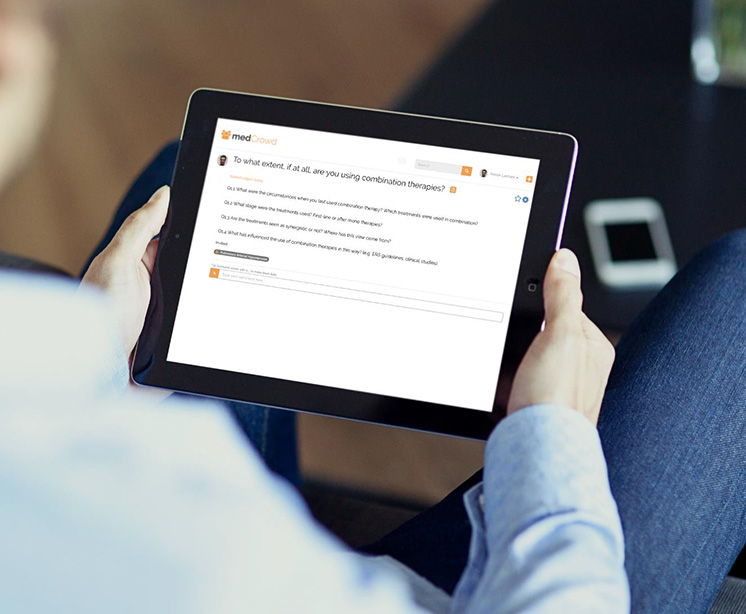
The instant messenger for health and care
Keeps everyone in touch
Secure conversations anytime and anywhere
Protects confidential information to the required health standards
You’ll be in good company










The medDigital Scientific Insight Team
Our talented Scientific Insight team at medDigital can help you gain scientific insights with medCrowd and a growing range of service offerings. The team are all scientists and can therefore help you gain the insights you need and help you understand the next steps too.
Learn moreScientific Insight Services
Digital Advisory Boards (DABs)
DABs enable you to engage with key stakeholders and discover insights quickly. HCPs interact and discuss the clinical landscape – remotely and asynchronously.
Clinical trial design & HTA submissions
Discover investigator insights from clinical development teams to optimise clinical trials, HTA submissions and NICE appraisals.
Online advice service
create a panel of expert advisors who can provide clinical advice as a digital service.
Patient panels
Talk directly with patients – learn about unmet, access to care, and their perspectives.
Market Research
We are BHBIA-certified scientific business intelligence professionals. Our team will lead focus groups, in-depth interviews and more, facilitating large qualitative data collection.
Academic focus groups
Bring together a group of academics to discuss and critically evaluate recent developments in basic or applied medical research.
Conference Support
We support you with conference management – from concept and content development, to post-event discussions.
Survey Tool
For quantitative data gathered through compliant closed question surveys. The Scientific Insight team can support with using our own or existing survey tools, track responses, and provide a summary report.
Find out more about our service offerings
Participant Benefits

Time & flexibility
Participants can connect whenever is most convenient for them, easily balancing their life and work with participation.

Multilingual capabilities
Participants can read & translate content without the concerns of language barriers, making it easier to participate than in a live environment. Our Scientific team will be able to translate your responses back into the main project language.

The best advice
Asynchronous conversations reduce time-pressure by giving participants more thinking time, allowing them to deliberate over questions, digest the information and craft carefully-considered responses.

Accessibility
Faster and more flexible than in-person advice-seeking activities, supporting science and patients by increasing access to important information.

Travel-free
Freedom to participate from any location eliminates the need for travel, supporting sustainability goals, but also allowing participants in more remote locations to participate seamlessly

Instant & simple
Our technology is simple and intuitive with the responsive medCrowd web app allowing contributions on the go.
Key Features
Convenient instant messaging
Asynchronous, text-based conversations
Connect globally across all time zones via laptop, desktop or mobile device
Unlimited number of conversations with participants
Open, close, and reopen conversations when you need
Share files for experts to review (whether videos, documents or images)
Tag, reply and react to each other’s messages
Add polls and links
Easily manage participation
Easily manage participation
Invite team members and participants via email or mobile number
Appoint additional admins to help
Withdraw comments or close conversations when needed
View invitation status and participation statistics
Approve online payments each month
Use the automated HSI Bot to facilitate pharmacovigilance reporting
Protects confidential information to the required standards
Data is encrypted at rest and in transit, meeting data protection standards.
Compliant with the NHS DSPT, HIPAA, UK Data Protection Act and the European General Data Protection Regulation with ISO 27001 certification being finalised.
Life Science Company Benefits

Insights
Participants have time to craft carefully-considered responses leading to more useful insights.

Diversity
Easily connecting with participants in different specialities, countries, and time zones allows you to draw from a diverse range of perspectives and advisors who may otherwise be too busy to travel.

Value for money
medCrowd provides a considerably less costly alternative to face-to-face engagements by absorbing the costs normally associated with travel & accommodation.

Time-saving
medCrowd projects can be set up rapidly, allowing for fast answers to questions to gain input from experts in real-time.

Data connections
medCrowd automatically collates and documents data which can also be integrated with software systems such as your customer relationship management (CRM) systems.

Environment
By eliminating unnecessary travel, medCrowd supports sustainability goals to protect the environment.
Life Science Company Feedback

Associate Director, Medical Affairs - Asynchronous DAB with a live Webinar (Synchronous)

Director, Global Medical Affairs – Asynchronous DAB

Global Events Team: Medical Affairs – Asynchronous DAB
Professional Feedback

Doctor, Nephrology, Russia

Doctor, Nephrology, Turkey